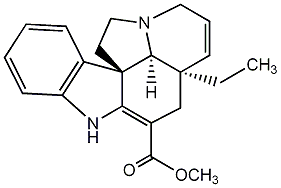
| Name |
Tabersonine |
| Formula |
C21H24N2O2 |
| Mw |
336.18377802 |
| CAS RN |
4429-63-4 |
| C_ID |
C00001773
, 
|
| InChIKey |
FNGGIPWAZSFKCN-SYDPFBBJNA-N |
| InChICode |
InChI=1S/C21H24N2O2/c1-3-20-9-6-11-23-12-10-21(19(20)23)15-7-4-5-8-16(15)22-17(21)14(13-20)18(24)25-2/h4-9,19,22H,3,10-13H2,1-2H3/t19-,20-,21-/m0/s1 |
| SMILES |
N1C2=C(C[C@]3([C@H]4[C@]2(c2c1cccc2)CCN4CC=C3)CC)C(=O)OC |
| Start Substs in Alk. Biosynthesis (Prediction) |
|
| Organism |
| Kingdom |
Family |
Species |
Reference |
| Plantae | Apocynaceae | Alstonia yunnanensis | Ref. |
| Plantae | Apocynaceae | Amsonia angustifolia | Ref. |
| Plantae | Apocynaceae | Amsonia illustris | Ref. |
| Plantae | Apocynaceae | Amsonia tabernaemontana | Ref. |
| Plantae | Apocynaceae | Catharanthus roseus  | Ref. |
| Plantae | Apocynaceae | Ervatamia divaricata Gouyahua | Ref. |
| Plantae | Apocynaceae | Vinca herbacea Waldst.et Kit.  | Ref. |
| Plantae | Apocynaceae | Voacanga africana  | Ref. |
|
|
zoom in
| Organism | Catharanthus roseus |
|---|
| Reference | Yin, et al., Modern Study of Chinese Drugs and Clinical Applications (1), Xueyuan Press, Beijing, (1993).
Sun, et al., Brief Handbook of Natural Active Compounds, Medicinal Science and Technology Press of China, Beijing, (1998) |
|---|
|