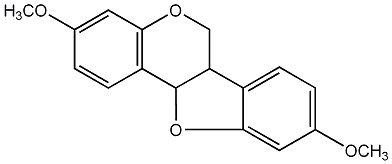
| Name |
(-)-Homopterocarpin
Baphinitone
3,9-Dimethoxypterocarpan |
| Formula |
C17H16O4 |
| Mw |
284.104859 |
| CAS RN |
606-91-7 |
| C_ID |
C00009613
, 
|
| InChIKey |
VPGIGLKLCFOWDN-UHFFFAOYNA-N |
| InChICode |
InChI=1S/C17H16O4/c1-18-10-4-6-13-15(7-10)20-9-14-12-5-3-11(19-2)8-16(12)21-17(13)14/h3-8,14,17H,9H2,1-2H3/t14-,17-/m1/s1 |
| SMILES |
c1(ccc2c(c1)OC[C@H]1[C@@H]2Oc2c1ccc(c2)OC)OC |
| Start Substs in Alk. Biosynthesis (Prediction) |
|
| Organism |
| Kingdom |
Family |
Species |
Reference |
| Plantae | Convolvulaceae | Erycibe expansa  | Ref. |
| Plantae | Fabaceae | Maackia amurensis  | Ref. |
| Plantae | Fabaceae | Ononis viscosa subsp. breviflora  | Ref. |
| Plantae | Fabaceae | Pericopsis angolensis  | Ref. |
| Plantae | Fabaceae | Pericopsis schliebenii | Ref. |
| Plantae | Fabaceae | Pterocarpus indicus  | Ref. |
| Plantae | Fabaceae | Pterocarpus spp. | Ref. |
| Plantae | Fabaceae | Swartzia madagascariensis  | Ref. |
| Plantae | Fabaceae | Trifolium hybridum  | Ref. |
|
|
zoom in
| Organism | Trifolium hybridum |
|---|
| Reference | Matsuda, et al., Planta Med, 70, (2004), 1201.
Buckingham(Executive Editor), Dictionary of Natural Products, Chapman & Hall, 1994, Vol1-7
1995, Vol8
1996, Vol9
1997, Vol10
1998, Vol11.
Sun, et al., Brief Handbook of Natural Active Compounds, Medicinal Science and Technology Press of China, Beijing, (1998).
Ji, et al., Pharmacological Action and Application of Available Antitumor Composition of Traditional Chinese Medicine, Heilongjiang Science and technology Press, Heilongjiang, (1998).
Harborne, The Handbook of Natural Flavonoids, 2, (1999), 517,Isoflavonoids and neoflavonoids
Cazeneuve,C.R.Acad.Sci.Paris,104,(1887),1722
Raudnitz,Ber.Dtsch.Chem.Ges.,68B,(1935),1862 |
|---|
|